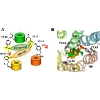
Activation processes of GPCRs
The existence of oligomeric assemblies of GPCRs has been confirmed by biochemical and biophysical studies including direct AFM imaging of rhodopsin in native membranes. The question is how they work together to transduce the signal into the cell and, further on, how the oligomeric state influences all aspects of their biological function. Crystallographic structure of rhodopsin revealed that its cytoplasmic surface is too small to bind the whole trimeric G protein and accommodate all interactions predicted from crosslinking data. According to our modeling, the trimeric transducin (G protein for rhodopsin) binds to rhodopsin tetramer.
We also revealed the modes of action of agonist/antagonist sensor in opioid receptors and investigated the central molecular switch in cannabinoid receptors (opioid and cannabinoid receptors are also GPCRs). We continue investigations of influence of ligands (agonists, antagonists and inverse agonists) on the receptor structure and the action of molecular switches in GPCRs.
- July 2023.GS-SMD web server
for SMD simulations of γ-secretase complex.
Publication in Nucleic Acids Research 2023 Web server issue.
- August 2022.COGRIMEN
- June 2021.GPCRsignalOur new service GPCRsignal was recently published in NAR 2021, W1.